Can you name a neglected tropical disease (NTD)? I would guess that most biologists can, or can at least make some accurate guesses. But by definition, these are maladies that don’t get adequate attention and resources, and so it is likely that many scientists are unaware of the current roster of NTDs. A new and exciting paper at Nature taught me that snakebite (envenoming, to be precise) is in fact a devastating NTD, with a heartbreaking death toll due in large part to the lack of effective and affordable* treatments. More importantly, the paper reports on how the authors discovered some simple and effective therapeutics with significant potential to make a difference. Their abstract is, to me, a tour de force of scientific writing that is straightforward and inspiring. (My favorite sentence suggests that we can “democratize therapeutic discovery.”) Here it is with references removed for readability:
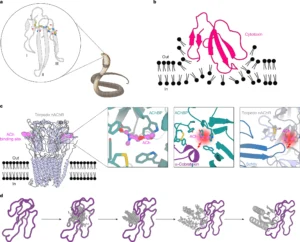
Snakebite envenoming remains a devastating and neglected tropical disease, claiming over 100,000 lives annually and causing severe complications and long-lasting disabilities for many more. Three-finger toxins (3FTx) are highly toxic components of elapid snake venoms that can cause diverse pathologies, including severe tissue damage and inhibition of nicotinic acetylcholine receptors, resulting in life-threatening neurotoxicity. At present, the only available treatments for snakebites consist of polyclonal antibodies derived from the plasma of immunized animals, which have high cost and limited efficacy against 3FTxs. Here we used deep learning methods to de novo design proteins to bind short-chain and long-chain α-neurotoxins and cytotoxins from the 3FTx family. With limited experimental screening, we obtained protein designs with remarkable thermal stability, high binding affinity and near-atomic-level agreement with the computational models. The designed proteins effectively neutralized all three 3FTx subfamilies in vitro and protected mice from a lethal neurotoxin challenge. Such potent, stable and readily manufacturable toxin-neutralizing proteins could provide the basis for safer, cost-effective and widely accessible next-generation antivenom therapeutics. Beyond snakebite, our results highlight how computational design could help democratize therapeutic discovery, particularly in resource-limited settings, by substantially reducing costs and resource requirements for the development of therapies for neglected tropical diseases.
*In a world in which a corrupt oligarch can buy the executive branch of the US government using less than 1% of his wealth, let’s not pretend that the main problem is “affordability.” The authors describe several other reasons why current approaches are badly inadequate.
De novo designed proteins neutralize lethal snake venom toxins
In Nature, 15 January 2025
From the groups of Timothy P. Jenkins and David Baker.
Snippet by Stephen Matheson
Image credit: Figure 1 from Vázquez Torres et al. linked above (CC-BY-NC-ND).